REDWOOD CITY, Calif. – June 29, 2018 – Pulmonx® Corp. announced today that the U.S. Food and Drug Administration (FDA) has approved the Zephyr® Endobronchial Valve System for treating severe emphysema patients. The Zephyr Valve is the first minimally-invasive device approved in the United States for treating patients with severe emphysema, a progressive and life-threatening form of chronic obstructive pulmonary disease (COPD).
The approval is based on positive clinical data from the pivotal LIBERATE Study and two other multicenter randomized control trials. In the LIBERATE study, patients treated with Zephyr Valves were able to breathe easier, be more active and energetic, be less short of breath, and enjoy a significantly improved quality of life compared to patients who received medical management alone.
As stated in the Summary of Safety and Effectiveness Data, the FDA granted the Zephyr Valve an expedited review because it “represents a breakthrough technology as the device offers bronchoscopic lung volume reduction without surgery and its associated risks. This device offers significant clinically meaningful advantage over the current standard of care and therefore its availability is also in the best interest of patients.”
“Zephyr Valves are a major step forward in treating severe emphysema patients who consistently feel short of breath despite all the medications we can offer. I have seen Zephyr Valve-treated patients getting back to a more active life doing the things they enjoy. As a physician, it is very gratifying to have a new treatment that can restore a patient’s confidence and change their life for the long term,” said Gerard Criner, MD, FACP, FACCP, Chair and Professor of Thoracic Medicine and Surgery, Lewis Katz School of Medicine at Temple University, and lead investigator for the LIBERATE Study.
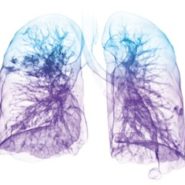
More than 15 million Americans suffer from COPD, and 3.5 million of those patients have emphysema.1 Despite using COPD medications, over one million emphysema patients continue to suffer symptoms of hyperinflation, in which air becomes trapped in the lungs and prevents new air from coming in, causing severe shortness of breath.2 The inability to get enough air often prevents these patients from doing simple daily activities, such as bathing, getting dressed, performing household chores and walking, without pausing to catch their breath. Until now, the only other options for these patients were highly invasive treatments such as lung volume reduction surgery or lung transplantation.
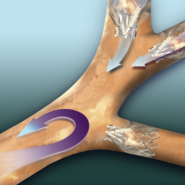
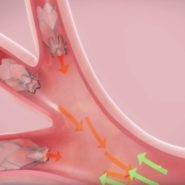
During a bronchoscopic procedure requiring no cutting or incisions, tiny Zephyr Valves are placed in the airways to occlude a diseased part of the lungs and reduce hyperinflation. This helps the healthier parts of the lungs to expand and lifts pressure off the diaphragm, thereby decreasing shortness of breath and making breathing easier.
Patients most likely to benefit from Zephyr Valve treatment can be identified with assessment tools also offered by Pulmonx. Physicians use the Pulmonx Chartis® Pulmonary Assessment System and StratX® Lung Analysis Platform to help identify potential responders to Zephyr Valve treatment.
“It is gratifying to be able to tell the many US patients who have contacted us that help is on the way. We thank FDA for its swift review of the Zephyr Valve. By combining the Zephyr Valves and our patient selection tools, we are bringing precision medicine to the treatment of severe emphysema,” said Pulmonx CEO Glen French.
Since 2007, more than 14,000 patients have been treated with the Zephyr Valve worldwide. Zephyr Valve treatment is included in emphysema treatment guidance issued by leading health organizations worldwide, including the Global Initiative for Chronic Obstructive Lung Disease (GOLD) and the UK’s National Institute for Health and Care Excellence (NICE).
The Pulmonx Zephyr® Endobronchial Valves are implantable bronchial valves indicated for the bronchoscopic treatment of adult patients with hyperinflation associated with severe emphysema in regions of the lung that have little to no collateral ventilation.
About Pulmonx
Based in Redwood City, California, and Neuchâtel, Switzerland, Pulmonx is the world leader in interventional pulmonology treatments for obstructive lung disease. For more information, visit www.MyLungsMyLife.com
- Centers for Disease Control and Prevention. Summary Health Statistics Tables for U.S. Adults: National Health Interview Survey 2016.https://www.cdc.gov/nchs/fastats/copd.htm. Accessed June 22, 2018.
- Soriano JB, Lamprecht B, Ramirez AS, et al. Mortality prediction in chronic obstructive pulmonary disease comparing the GOLD 2007 and 2011 staging systems: a pooled analysis of individual patient data. Lancet Respir Med. 2015;3(6):443–50.